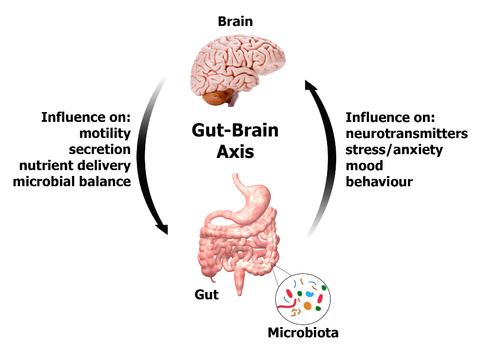
Who Knew We Had Three Brains?
The gut has its own independent nervous system, known as the enteric nervous system, that consists of about 500 million neurons. This system is embedded in the lining of the gastrointestinal tract, beginning at the esophagus and ending at the anus. Amazingly, the enteric nervous system can operate independently from the brain – it can function without it, and has been dubbed our “second brain”. It has a two way communication with the brain and central nervous system via the parasympathetic fibers of the vagus nerve and the sympathetic fibers of the prevertebral ganglia. This channel is known as the gut-brain axis, and links the emotional and cognitive centres of the brain with intestinal function, and vice-versa.
The gut-brain axis is modulated by neurotransmitters, hormones, immune system cytokines, as well as the organisms present in the intestines, what you eat, and the overall health of the gut. The enteric nervous system affects what happens in the rest of the body, including the brain. It contributes to our level of anxiety, appetite, satiety, our mood, emotions, learning, memory and overall inflammation. All of these factors play a role in mental health and susceptibility to addiction. If there is infection or inflammation in the gut, the brain will get the message, and may react with symptoms of anxiety, depression or other mental illness.
The enteric nervous system makes use of 30 different neurotransmitters, including acetylcholine (memory), dopamine (motivation) and serotonin (happiness). More than 90% of the body’s serotonin and 50% of its dopamine are synthesized in the gut, regulated by the gut flora.
And what is the third brain, you ask? The heart – it has over over 40,000 neurons that can work independently from the brain, giving it the ability to sense, process information, make decisions, and demonstrate a type of learning and memory. It secretes several hormones, including oxytocin, known as the love or “bonding” hormone.
Gut Health and Relapse in Alcoholics
Research suggests that bacteria in the gut may play a role in alcohol addiction and the likelihood of relapse after rehab. Alcohol addiction is often associated with an imbalance in the intestinal flora. One study found that 26 out of 60 alcoholics suffered from leaky gut syndrome and had a low amount of a specific intestinal bacteria, Faecalibacterium prausnitzii, which has anti-inflammatory properties. Leaky gut syndrome is linked to inflammation of the gut and diseases like Crohn’s disease, food allergies, asthma and arthritis. After 19 days without alcohol the 26 alcoholics with leaky gut still scored high on tests that measured depression, anxiety, and alcohol cravings.
In contrast, the remaining 34 subjects with normal gut flora recovered faster, measuring lower on the same tests. Their scores decreased to levels comparable with a control group who didn’t have a drinking problem.[i]
Chronic alcohol ingestion also increases the risk of colon cancer, as bacteria in the colon and rectum convert ethanol to acetaldehyde, which is a carcinogen.
Mental Illness and Gut Dysbiosis
Mental illness such as bipolar, depression, anxiety, and psychotic disorders are not only caused by trauma, physical or emotional stress, nutritional deficit, infection, hormonal imbalance and/or genetic susceptibility, but can be inflammatory conditions related to the function of the immune system and the health of the gut.[ii] [iii] [iv] Over 500 species of bacteria comprise the gut microbiome, consisting of about 100 trillion organisms.
Social and psychological stressors affect not only the immune system, but also the bacterial composition of the gut, favoring the growth of species that promote inflammation.[v] These inflammatory species have a direct connection with the brain via the neurotransmitters they produce and the vagus nerve receptors in the gut.[vi] The vagus nerve seems to be capable of differentiating between harmless and potentially harmful bacteria, even when there is no inflammation, and vagal pathways can send signals to the brain that can induce either anxiety-producing or calming effects.
Probiotics to the Rescue
Probiotics such as Lactobacillus and Bifidobacteria are anti-inflammatory, and can downplay the damaging immune and stress response caused by unfavourable bacteria or life events. Demonstrations in both animal and human studies have shown that the administration of beneficial bacteria can reduce both inflammation and anxiety or behavioral signs of distress. [vii] [viii] [ix] They can be considered as bacterial anti-depressants.
One small study demonstrated that infants who were supplemented with probiotics had a much lower rate of ADHD or autistic spectrum disorder when they grew up to be young teenagers, compared to a control group who were not given probiotics as infants.[x]
Another study found that depressed individuals had a higher population of Bacteroidales species in the gut.[xi]
The study of the relationship between gut bacteria, mental health and addiction is still in its infancy. We do know however, that many people suffering from anxiety, depression and addiction have disordered gut flora and can benefit from taking a probiotic containing various strains of Lactobacillus and Bifidobacteria.
Sat Dharam Kaur ND is a naturopathic doctor practicing in Owen Sound, Ontario. She developed the Beyond Addiction program and teaches internationally.
For upcoming Beyond Addiction programs see https://beyondaddiction.ca/events/training-programs/
References:
[i] Leclercq S, Matamoros S, Cani PD et al. Intestinal permeability, gut-bacterial dysbiosis, and behavioral markers of alcohol-dependence severity. Proc Nat Acad Sci USA vol III no. 42. Sept 15, 2014
[ii] Berk M, Williams LJ, Jacka FN, O’Neil A, Pasco JA, Moylan S, Allen NB, Stuart AL, Hayley AC, Byrne ML, Maes M. So depression is an inflammatory disease, but where does the inflammation come from? BMC Med. 2013
[iii] Leonard B, Maes M. Mechanistic explanations how cell-mediated immune activation, inflammation and oxidative and nitrosative stress pathways and their sequels and concomitants play a role in the pathophysiology of unipolar depression. Neurosci Biobehav Rev. 2012
[iv] Kapczinski F, Dal-Pizzol F, Teixeira AL, Magalhaies PV, Kaur-Sant’Anna M, Klamt F, Moreira JC, de Bittencourt Pasquali MA, Fries GR, Quevedo J, Gama CS, Post R. Peripheral biomarkers and illness activity in bipolar disorder. J Psychiatr Res. 2011
[v] Bailey MT, Dowd SE, Galley JD, Hufnagle AR, Allen RG, Lyte M. Exposure to a social stressor alters the structure of the intestinal microbiota: implications for stressor-induced immunomodulation. Brain Behav Immun. 2011
[vi] Forsythe P, Bienenstock J, Kunze WA. Vagal pathways for microbiome-brain-gut axis communication.Adv Exp Med Biol. 2014
[vii] Cryan JF, Dinan TG. Mind-altering microorganisms: the impact of the gut microbiota on brain and behaviour. Nat Rev Neurosci. 2012
[viii] Messaoudi M, Lalonde R, Violle N, Javelot H, Desor D, Nejdi A, Bisson JF, Rougeot C, Pichelin M, Cazaubiel M, Cazaubiel JM. Assessment of psychotropic-like properties of a probiotic formulation (Lactobacillus helveticus R0052 and Bifidobacterium longum R0175) in rats and human subjects. Br J Nutr. 2011
[ix] Tillisch K, Labus J, Kilpatrick L, Jiang Z, Stains J, Ebrat B, Guyonnet D, Legrain-Raspaud S, Trotin B, Naliboff B, Mayer EA. Consumption of fermented milk product with probiotic modulates brain activity.Gastroenterology. 2013
[x] Pärtty A, Kalliomäki M, Wacklin P, Salminen S, Isolauri E. A possible link between early probiotic intervention and the risk of neuropsychiatric disorders later in childhood: a randomized trial. Pediatr Res. 2015
[xi] Naseribafrouei A, Hestad K, Avershina E, Sekelja M, Linløkken A, Wilson R, Rudi K. Correlation between the human fecal microbiota and depression. Neurogastroenterol Motil. 2014

Thank you for this excellent informative article. It is something that I’ve firmly believed in for a very longtime. All practicing physicians of all types and specialities must be familiar with it.
Wow how great is this Thank you.
I attend “Relapse Prevention Group” for alcohol and had been led to ‘Mindfulness’ & so in my orecious one-2-one session
My worker mapped out a diagram of the brain and showed me how the brain
And the breath work in letting Go
“BUT!”-I asked her;”Whaf about the
GUT….What is going on In Here!.
I pointed helplessly to my Solar Plexus where I feel
All the “Less than worthy feelings. My fears of alcohol and drinking again.
She told me::”No it is the brain.”.
I have a session in 2 days with her.
I am going to take this in to show her
As she plays a prominent role in helping others.
She said she found me great as I was so open to learning. I said if was due to my nursing background bug work as a Reflexologist/healer too maybe.
I’m used to playing small..my theme of survival or rather “just existing” but not really fully ever entering into the not of holding All that I am.
So I’m really excited about this article
I know as a Reflexologist the connections of the “biofeedback mechanism” throughout the whole body. We would start on the brain to quieter the pituitary or “Master Gland” and that is then calming the endocrines.
Off to PRINT IT OFF now.
I’m hoping the course comes to the UK in 11/17. I need help to overcome the deep beliefs that hold me a prisoner.
Thank you So very very much.
The body and mind and spirit are all so linked. Extraordinary really.
Interested in more info